-
 Factors Associated with the Implementation of Pediatric Immunization Services: A Survey of Community Pharmacies
Factors Associated with the Implementation of Pediatric Immunization Services: A Survey of Community Pharmacies -
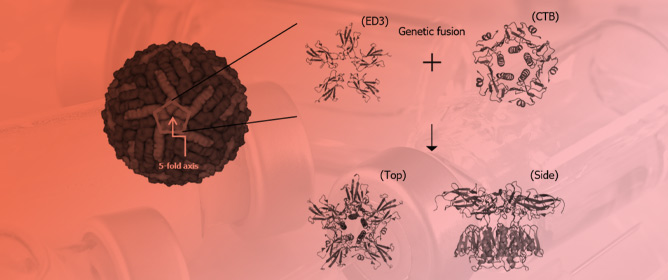 Harnessing Pentameric Scaffold of Cholera Toxin B (CTB) for Design of Subvirion Recombinant Dengue Virus Vaccine
Harnessing Pentameric Scaffold of Cholera Toxin B (CTB) for Design of Subvirion Recombinant Dengue Virus Vaccine -
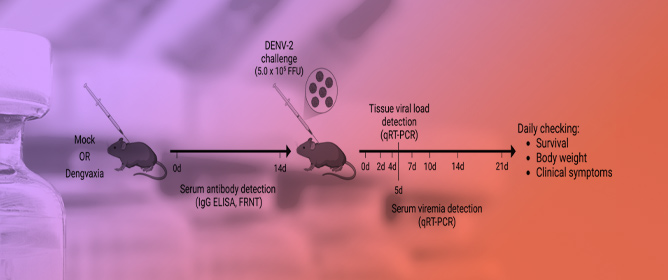 A Novel, Comprehensive A129 Mouse Model for Investigating Dengue Vaccines and Evaluating Pathogenesis
A Novel, Comprehensive A129 Mouse Model for Investigating Dengue Vaccines and Evaluating Pathogenesis -
 Do Not Leave Your Patients in the Dark—Vaccination Recommendations in Rheumatological Patients
Do Not Leave Your Patients in the Dark—Vaccination Recommendations in Rheumatological Patients
Journal Description
Vaccines
Vaccines
is an international, peer-reviewed, open access journal published monthly online by MDPI. The American Society for Virology (ASV) is affiliated with Vaccines and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q1 (Immunology) / CiteScore - Q1 (Pharmacology (medical))
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 19.2 days after submission; acceptance to publication is undertaken in 2.9 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
7.8 (2022);
5-Year Impact Factor:
7.4 (2022)
Latest Articles
A Proof-of-Concept Study to Develop a Peptide-Based Vaccine against Salmon Lice Infestation in Atlantic Salmon (Salmo salar L.)
Vaccines 2024, 12(5), 456; https://doi.org/10.3390/vaccines12050456 - 24 Apr 2024
Abstract
Proteins present in blood samples from Atlantic salmon (Salmo salar) infected with salmon lice (Lepeophtheirus salmonis) were analyzed using liquid chromatography–high-resolution mass spectrometry. Bioinformatic analyses revealed 1820 proteins, of which 58 were assigned to lice. Among these, peroxiredoxin-2, an
[...] Read more.
Proteins present in blood samples from Atlantic salmon (Salmo salar) infected with salmon lice (Lepeophtheirus salmonis) were analyzed using liquid chromatography–high-resolution mass spectrometry. Bioinformatic analyses revealed 1820 proteins, of which 58 were assigned to lice. Among these, peroxiredoxin-2, an antioxidant protein, was found relevant with respect to blood feeding of the parasite. The three-dimensional structure analysis of the protein revealed a surface amino acid sequence of interest. A 13-amino-acid peptide was selected as a potential antigen due to its predicted solubility, antigenicity, probable non-allergenic, and non-toxic nature. This peroxiredoxin-2-derived peptide was synthesized, combined with a commercially available adjuvant, and used for vaccination. The test vaccine demonstrated a 60–70% protection rate against early-stage Lepeophtheirus salmonis infection in a challenge trial in Norway. Additionally, the vaccine was tested against salmon lice (Caligus rogercresseyi) in Chile, where a remarkable 92% reduction in the number of adult lice was observed. Thus, in combination with the selected adjuvant, the peptide showed antigenic potential, making it a suitable candidate for future vaccine development. The approach described holds promise for the development of peptide vaccines against various ectoparasites feeding on blood or skin secretions of their hosts.
Full article
Open AccessArticle
Advax-SM™-Adjuvanted COBRA (H1/H3) Hemagglutinin Influenza Vaccines
by
Pedro L. Sanchez, Greiciely Andre, Anna Antipov, Nikolai Petrovsky and Ted M. Ross
Vaccines 2024, 12(5), 455; https://doi.org/10.3390/vaccines12050455 - 24 Apr 2024
Abstract
Adjuvants enhance immune responses stimulated by vaccines. To date, many seasonal influenza vaccines are not formulated with an adjuvant. In the present study, the adjuvant Advax-SM™ was combined with next generation, broadly reactive influenza hemagglutinin (HA) vaccines that were designed using a computationally
[...] Read more.
Adjuvants enhance immune responses stimulated by vaccines. To date, many seasonal influenza vaccines are not formulated with an adjuvant. In the present study, the adjuvant Advax-SM™ was combined with next generation, broadly reactive influenza hemagglutinin (HA) vaccines that were designed using a computationally optimized broadly reactive antigen (COBRA) methodology. Advax-SM™ is a novel adjuvant comprising inulin polysaccharide and CpG55.2, a TLR9 agonist. COBRA HA vaccines were combined with Advax-SM™ or a comparator squalene emulsion (SE) adjuvant and administered to mice intramuscularly. Mice vaccinated with Advax-SM™ adjuvanted COBRA HA vaccines had increased serum levels of anti-influenza IgG and IgA, high hemagglutination inhibition activity against a panel of H1N1 and H3N2 influenza viruses, and increased anti-influenza antibody secreting cells isolated from spleens. COBRA HA plus Advax-SM™ immunized mice were protected against both morbidity and mortality following viral challenge and, at postmortem, had no detectable lung viral titers or lung inflammation. Overall, the Advax-SM™-adjuvanted COBRA HA formulation provided effective protection against drifted H1N1 and H3N2 influenza viruses.
Full article
(This article belongs to the Special Issue Advances in Influenza Virus Vaccines)
►▼
Show Figures
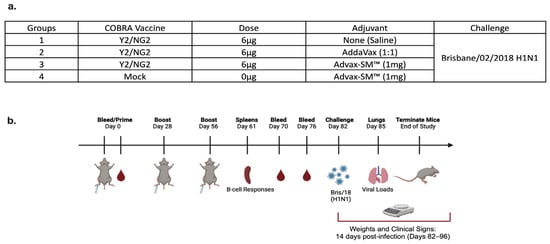
Figure 1
Open AccessArticle
Evolving Altruistic Attitudes towards Vaccination Post COVID-19 Pandemic: A Comparative Analysis across Age Groups
by
Verena Barbieri, Christian J. Wiedermann, Stefano Lombardo, Giuliano Piccoliori, Timon Gärtner and Adolf Engl
Vaccines 2024, 12(5), 454; https://doi.org/10.3390/vaccines12050454 - 24 Apr 2024
Abstract
Altruism plays an essential role in promoting vaccine uptake, an issue that came to the fore during the COVID-19 pandemic through discussions of herd immunity and altruistic motivations. In response, the primary objective of this cross-sectional survey was to explore how altruistic attitudes
[...] Read more.
Altruism plays an essential role in promoting vaccine uptake, an issue that came to the fore during the COVID-19 pandemic through discussions of herd immunity and altruistic motivations. In response, the primary objective of this cross-sectional survey was to explore how altruistic attitudes have evolved in the post-pandemic era and to assess their effectiveness in motivating vaccination behavior in different age groups. The study aimed to elucidate changes in altruistic motivations for vaccination and their implications for public health strategies. Using a representative sample of the adult population of South Tyrol, Italy, including 1388 participants, altruism was assessed in 2023 with the scales of the Elderly Care Research Center (ECRC) and the International Personality Item Pool (IPIP) subscale of the version 5F30F-R1. Its association with demographic variables, vaccination attitudes and personal beliefs in two age groups (18–69 years, 70+ years) was analyzed. The results reveal distinct predictors of altruism across these scales and age groups, suggesting a shift in altruistic attitudes towards vaccination when comparing data from a similar survey conducted in 2021 with the 2023 results. Consequently, the use of altruism scales for different age groups is warranted. This study highlights the need for further research in this field. It concludes that while promoting altruistic behavior to increase vaccine uptake appears to be effective primarily among the younger population, emphasizing personal safety is more appropriate for encouraging vaccination among older individuals.
Full article
(This article belongs to the Special Issue COVID-19 Vaccine Acceptance and Uptake: Insights from Behavioural and Social Sciences)
►▼
Show Figures
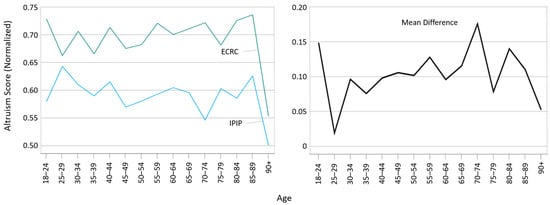
Figure 1
Open AccessArticle
COVID-19 Vaccine Effectiveness among Patients with Psoriatic Disease: A Population-Based Study
by
Tal Gazitt, Lihi Eder, Walid Saliba, Nili Stein, Ilan Feldhamer, Arnon Dov Cohen and Devy Zisman
Vaccines 2024, 12(5), 453; https://doi.org/10.3390/vaccines12050453 - 24 Apr 2024
Abstract
Limited information is available on the effectiveness of COVID-19 vaccination in patients with psoriasis and psoriatic arthritis (psoriatic disease (PsD)). The objective of our research was to assess the effectiveness of mRNA COVID-19 vaccination in preventing SARS-CoV-2 positivity and severe infection in a
[...] Read more.
Limited information is available on the effectiveness of COVID-19 vaccination in patients with psoriasis and psoriatic arthritis (psoriatic disease (PsD)). The objective of our research was to assess the effectiveness of mRNA COVID-19 vaccination in preventing SARS-CoV-2 positivity and severe infection in a cohort of patients with PsD and the association of immunosuppressants on SARS-CoV-2 infection-related outcomes from December 2020 to December 2021. Vaccine effectiveness was assessed in a matched nested case control study using conditional logistic regression adjusted for demographics, comorbidities and immunosuppressant use. Study outcomes included SARS-CoV-2 positivity and severe COVID-19 (moderate-to-severe COVID-19-related hospitalizations or death). At least one dose of mRNA COVID-19 vaccine was associated with reduced risk of SARS-CoV-2 positivity and severe COVID-19 (OR = 0.41 (95% CI, 0.38–0.43) and OR = 0.15 (95% CI, 0.11–0.20), respectively). A more significant effect was found among patients who received three vaccines doses compared with those who did not receive any (OR (for positive SARS-CoV-2) = 0.13 (95% CI, 0.12–0.15) and OR (for severe disease) = 0.02 (0.01–0.05)). Etanercept and methotrexate were associated with higher risk of SARS-CoV-2 positivity (1.58 (1.19–2.10), p = 0.001 and 1.25 (1.03–1.51), p = 0.03, respectively). In conclusion, our results show that mRNA COVID-19 vaccines are effective in reducing both infection and severe COVID-19-related outcomes.
Full article
(This article belongs to the Section COVID-19 Vaccines and Vaccination)
►▼
Show Figures
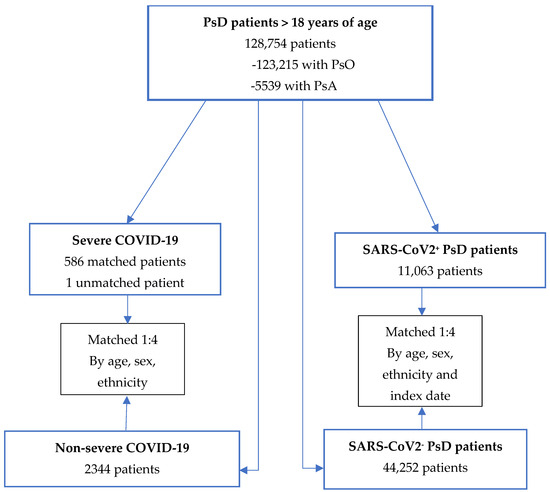
Figure 1
Open AccessArticle
BCG Vaccination-Associated Lower HbA1c and Increased CD25 Expression on CD8+ T Cells in Patients with Type 1 Diabetes in Ghana
by
Wilfred Aniagyei, Sumaya Mohayideen, Osei Sarfo-Kantanka, Sarah Bittner, Monika M. Vivekanandan, Joseph F. Arthur, Agnes O. Boateng, Augustine Yeboah, Hubert S. Ahor, Shadrack O. Asibey, Elizabeth Owusu, Diran Herebian, Maximilian Huttasch, Volker Burkart, Robert Wagner, Michael Roden, Ernest Adankwah, Dorcas O. Owusu, Ertan Mayatepek, Marc Jacobsen, Richard O. Phillips and Julia Seyfarthadd
Show full author list
remove
Hide full author list
Vaccines 2024, 12(5), 452; https://doi.org/10.3390/vaccines12050452 - 24 Apr 2024
Abstract
BCG vaccination affects other diseases beyond tuberculosis by unknown—potentially immunomodulatory—mechanisms. Recent studies have shown that BCG vaccination administered during overt type 1 diabetes (T1D) improved glycemic control and affected immune and metabolic parameters. Here, we comprehensively characterized Ghanaian T1D patients with or without
[...] Read more.
BCG vaccination affects other diseases beyond tuberculosis by unknown—potentially immunomodulatory—mechanisms. Recent studies have shown that BCG vaccination administered during overt type 1 diabetes (T1D) improved glycemic control and affected immune and metabolic parameters. Here, we comprehensively characterized Ghanaian T1D patients with or without routine neonatal BCG vaccination to identify vaccine-associated alterations. Ghanaian long-term T1D patients (n = 108) and matched healthy controls (n = 214) were evaluated for disease-related clinical, metabolic, and immunophenotypic parameters and compared based on their neonatal BCG vaccination status. The majority of study participants were BCG-vaccinated at birth and no differences in vaccination rates were detected between the study groups. Notably, glycemic control metrics, i.e., HbA1c and IDAA1c, showed significantly lower levels in BCG-vaccinated as compared to unvaccinated patients. Immunophenotype comparisons identified higher expression of the T cell activation marker CD25 on CD8+ T cells from BCG-vaccinated T1D patients. Correlation analysis identified a negative correlation between HbA1c levels and CD25 expression on CD8+ T cells. In addition, we observed fractional increases in glycolysis metabolites (phosphoenolpyruvate and 2/3-phosphoglycerate) in BCG-vaccinated T1D patients. These results suggest that neonatal BCG vaccination is associated with better glycemic control and increased activation of CD8+ T cells in T1D patients.
Full article
(This article belongs to the Special Issue Immunity and Vaccination against Bacterial Infections)
►▼
Show Figures
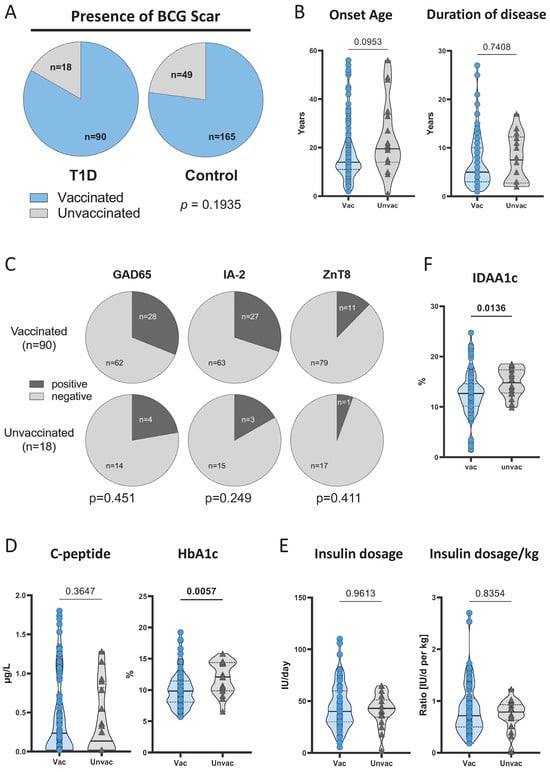
Figure 1
Open AccessArticle
Immunogenicity and Protective Efficacy of Psoralen-Inactivated SARS-CoV-2 Vaccine in Nonhuman Primates
by
John W. Sanders, Daniel Ewing, Appavu K. Sundaram, Christopher Scott Gamble, Maria Blevins, Zhaodong Liang, Leigh Ann Sanders, David A. Ornelles, Peifang Sun, Klara Lenart, Hendrik Feuerstein, Karin Loré, Nikolai Petrovsky, Maya Williams and Kevin R. Porter
Vaccines 2024, 12(5), 451; https://doi.org/10.3390/vaccines12050451 - 24 Apr 2024
Abstract
COVID-19 caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has significantly impacted public health and the economy worldwide. Most of the currently licensed COVID-19 vaccines act by inhibiting the receptor-binding function of the SARS-CoV-2 spike protein. The constant emergence of SARS-CoV-2 variants
[...] Read more.
COVID-19 caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has significantly impacted public health and the economy worldwide. Most of the currently licensed COVID-19 vaccines act by inhibiting the receptor-binding function of the SARS-CoV-2 spike protein. The constant emergence of SARS-CoV-2 variants resulting from mutations in the receptor-binding domain (RBD) leads to vaccine immune evasion and underscores the importance of broadly acting COVID-19 vaccines. Inactivated whole virus vaccines can elicit broader immune responses to multiple epitopes of several antigens and help overcome such immune evasions. We prepared a psoralen-inactivated SARS-CoV-2 vaccine (SARS-CoV-2 PsIV) and evaluated its immunogenicity and efficacy in nonhuman primates (NHPs) when administered with the Advax-CpG adjuvant. We also evaluated the SARS-CoV-2 PsIV as a booster shot in animals vaccinated with a DNA vaccine that can express the full-length spike protein. The Advax-CpG-adjuvanted SARS-CoV-2 PsIV elicited a dose-dependent neutralizing antibody response in the NHPs, as measured using a serum microneutralization assay against the SARS-CoV-2 Washington strain and the Delta variant. The animals vaccinated with the DNA vaccine followed by a boosting dose of the SARS-CoV-2 PsIV exhibited the highest neutralizing antibody responses and were able to quickly clear infection after an intranasal challenge with the SARS-CoV-2 Delta variant. Overall, the data show that the Advax-CpG-adjuvanted SARS-CoV-2 PsIV, either by itself or as a booster shot following nucleic acid (NA) vaccines, has the potential to protect against emerging variants.
Full article
(This article belongs to the Special Issue COVID Vaccines: Design, Development, and Immune Response Studies)
►▼
Show Figures

Figure 1
Open AccessArticle
Prospective and Cross-Sectional Factors Predicting Caregiver Motivation to Vaccinate Children with Attention-Deficit/Hyperactivity Disorder against COVID-19: A Follow-Up Study
by
Tai-Ling Liu, Ray C. Hsiao, Wen-Jiun Chou and Cheng-Fang Yen
Vaccines 2024, 12(5), 450; https://doi.org/10.3390/vaccines12050450 - 23 Apr 2024
Abstract
Adolescents with attention-deficit/hyperactivity disorder (ADHD) have higher risks of contracting COVID-19 and worse outcomes compared with adolescents without ADHD. The most effective method of preventing infection is vaccination. This follow-up study explored the prospective and cross-sectional factors influencing caregiver willingness to vaccinate children
[...] Read more.
Adolescents with attention-deficit/hyperactivity disorder (ADHD) have higher risks of contracting COVID-19 and worse outcomes compared with adolescents without ADHD. The most effective method of preventing infection is vaccination. This follow-up study explored the prospective and cross-sectional factors influencing caregiver willingness to vaccinate children with ADHD against COVID-19. Baseline data on caregiver demographics, affiliate stigma, parenting stress, emotional difficulties, beliefs regarding the causes of ADHD, and ADHD symptoms were collected prior to the outbreak of the COVID-19 pandemic in Taiwan. At follow-up, the study assessed caregiver willingness to vaccinate children with ADHD, the challenges caregivers faced in parenting during the pandemic, and ADHD symptoms. The results revealed that caregiver age at baseline was positively associated with a willingness to vaccinate children against COVID-19 at follow-up. By contrast, the belief that ADHD resulted from failures in parental discipline at baseline was negatively associated with caregiver willingness to vaccinate. Parenting challenges were also negatively associated with caregiver willingness to vaccinate. Therefore, the age of caregivers, beliefs about the causes of ADHD, and parenting challenges during the pandemic should be considered when developing interventions to enhance caregiver willingness to vaccinate children with ADHD.
Full article
(This article belongs to the Special Issue COVID-19 Vaccine Acceptance and Uptake: Insights from Behavioural and Social Sciences)
Open AccessArticle
Polymeric Caffeic Acid Acts as an Antigen Delivery Carrier for Mucosal Vaccine Formulation by Forming a Complex with an Antigenic Protein
by
Rui Tada, Yuzuho Nagai, Miki Ogasawara, Momoko Saito, Akihiro Ohshima, Daisuke Yamanaka, Jun Kunisawa, Yoshiyuki Adachi and Yoichi Negishi
Vaccines 2024, 12(5), 449; https://doi.org/10.3390/vaccines12050449 - 23 Apr 2024
Abstract
The development of mucosal vaccines, which can generate antigen-specific immune responses in both the systemic and mucosal compartments, has been recognized as an effective strategy for combating infectious diseases caused by pathogenic microbes. Our recent research has focused on creating a nasal vaccine
[...] Read more.
The development of mucosal vaccines, which can generate antigen-specific immune responses in both the systemic and mucosal compartments, has been recognized as an effective strategy for combating infectious diseases caused by pathogenic microbes. Our recent research has focused on creating a nasal vaccine system in mice using enzymatically polymerized caffeic acid (pCA). However, we do not yet understand the molecular mechanisms by which pCA stimulates antigen-specific mucosal immune responses. In this study, we hypothesized that pCA might activate mucosal immunity at the site of administration based on our previous findings that pCA possesses immune-activating properties. However, contrary to our initial hypothesis, the intranasal administration of pCA did not enhance the expression of various genes involved in mucosal immune responses, including the enhancement of IgA responses. Therefore, we investigated whether pCA forms a complex with antigenic proteins and enhances antigen delivery to mucosal dendritic cells located in the lamina propria beneath the mucosal epithelial layer. Data from gel filtration chromatography indicated that pCA forms a complex with the antigenic protein ovalbumin (OVA). Furthermore, we examined the promotion of OVA delivery to nasal mucosal dendritic cells (mDCs) after the intranasal administration of pCA in combination with OVA and found that OVA uptake by mDCs was increased. Therefore, the data from gel filtration chromatography and flow cytometry imply that pCA enhances antigen-specific antibody production in both mucosal and systemic compartments by serving as an antigen-delivery vehicle.
Full article
(This article belongs to the Special Issue Advance in Nanoparticles as Vaccine Adjuvants)
►▼
Show Figures
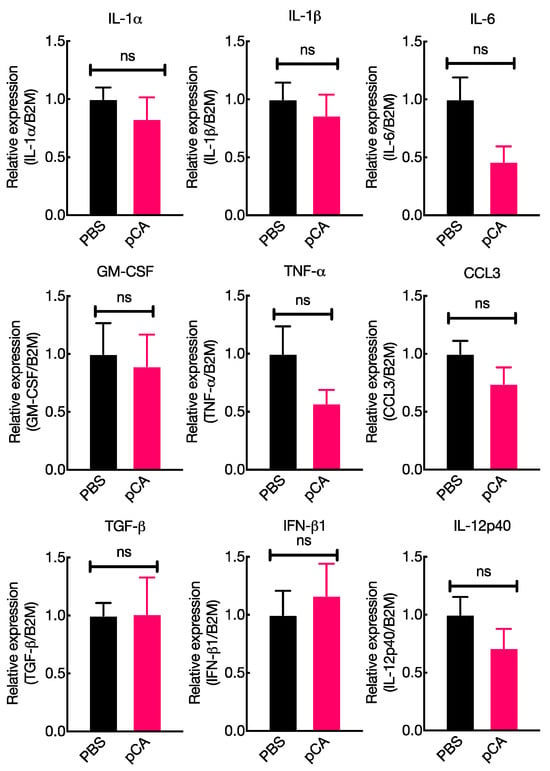
Figure 1
Open AccessArticle
Development of a Candidate TMV Epitope Display Vaccine against SARS-CoV-2
by
Kelvin Phiri and Larry Grill
Vaccines 2024, 12(5), 448; https://doi.org/10.3390/vaccines12050448 - 23 Apr 2024
Abstract
Essential in halting the COVID-19 pandemic caused by SARS-CoV-2, it is crucial to have stable, effective, and easy-to-manufacture vaccines. We developed a potential vaccine using a tobacco mosaic virus (TMV) epitope display model presenting peptides derived from the SARS-CoV-2 spike protein. The TMV-epitope
[...] Read more.
Essential in halting the COVID-19 pandemic caused by SARS-CoV-2, it is crucial to have stable, effective, and easy-to-manufacture vaccines. We developed a potential vaccine using a tobacco mosaic virus (TMV) epitope display model presenting peptides derived from the SARS-CoV-2 spike protein. The TMV-epitope fusions in laboratory tests demonstrated binding to the SARS-CoV-2 polyclonal antibodies. The fusion constructs maintained critical epitopes of the SARS-CoV-2 spike protein, and two in particular spanned regions of the receptor-binding domain that have mutated in the more recent SARS-CoV-2 variants. This would allow for the rapid modification of vaccines in response to changes in circulating variants. The TMV-peptide fusion constructs also remained stable for over 28 days when stored at temperatures between −20 and 37 °C, an ideal property when targeting developing countries. Immunogenicity studies conducted on BALB/c mice elicited robust antibody responses against SARS-CoV-2. A strong IFNγ response was also observed in immunized mice. Three of the six TMV-peptide fusion constructs produced virus-neutralizing titers, as measured with a pseudovirus neutralization assay. These TMV-peptide fusion constructs can be combined to make a multivalent vaccine that could be adapted to meet changing virus variants. These findings demonstrate the development of a stable COVID-19 vaccine candidate by combining SARS-CoV-2 spike protein-derived peptides presented on the surface of a TMV nanoparticle.
Full article
(This article belongs to the Special Issue COVID Vaccines: Design, Development, and Immune Response Studies)
Open AccessArticle
Correlates of Breakthrough SARS-CoV-2 Infections in People with HIV: Results from the CIHR CTN 328 Study
by
Cecilia T. Costiniuk, Terry Lee, Joel Singer, Yannick Galipeau, Corey Arnold, Marc-André Langlois, Judy Needham, Mohammad-Ali Jenabian, Ann N. Burchell, Hasina Samji, Catharine Chambers, Sharon Walmsley, Mario Ostrowski, Colin Kovacs, Darrell H. S. Tan, Marianne Harris, Mark Hull, Zabrina L. Brumme, Hope R. Lapointe, Mark A. Brockman, Shari Margolese, Enrico Mandarino, Suzanne Samarani, Bertrand Lebouché, Jonathan B. Angel, Jean-Pierre Routy, Curtis L. Cooper and Aslam H. Anisadd
Show full author list
remove
Hide full author list
Vaccines 2024, 12(5), 447; https://doi.org/10.3390/vaccines12050447 - 23 Apr 2024
Abstract
COVID-19 breakthrough infection (BTI) can occur despite vaccination. Using a multi-centre, prospective, observational Canadian cohort of people with HIV (PWH) receiving ≥2 COVID-19 vaccines, we compared the SARS-CoV-2 spike (S) and receptor-binding domain (RBD)-specific IgG levels 3 and 6 months post second dose,
[...] Read more.
COVID-19 breakthrough infection (BTI) can occur despite vaccination. Using a multi-centre, prospective, observational Canadian cohort of people with HIV (PWH) receiving ≥2 COVID-19 vaccines, we compared the SARS-CoV-2 spike (S) and receptor-binding domain (RBD)-specific IgG levels 3 and 6 months post second dose, as well as 1 month post third dose, in PWH with and without BTI. BTI was defined as positivity based on self-report measures (data up to last study visit) or IgG data (up to 1 month post dose 3). The self-report measures were based on their symptoms and either a positive PCR or rapid antigen test. The analysis was restricted to persons without previous COVID-19 infection. Persons without BTI remained COVID-19-naïve until ≥3 months following the third dose. Of 289 participants, 92 developed BTI (31.5 infections per 100 person-years). The median days between last vaccination and BTI was 128 (IQR 67, 176), with the most cases occurring between the third and fourth dose (n = 59), corresponding to the Omicron wave. In analyses adjusted for age, sex, race, multimorbidity, hypertension, chronic kidney disease, diabetes and obesity, a lower IgG S/RBD (log10 BAU/mL) at 1 month post dose 3 was significantly associated with BTI, suggesting that a lower IgG level at this time point may predict BTI in this cohort of PWH.
Full article
(This article belongs to the Special Issue COVID-19 Vaccines and Immune Response)
►▼
Show Figures
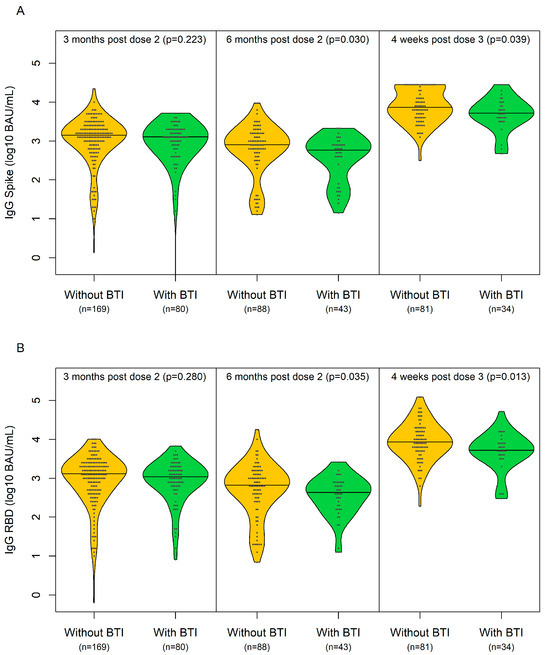
Figure 1
Open AccessArticle
Single Ferritin Nanocages Expressing SARS-CoV-2 Spike Variants to Receptor and Antibodies
by
Monikaben Padariya and Umesh Kalathiya
Vaccines 2024, 12(5), 446; https://doi.org/10.3390/vaccines12050446 - 23 Apr 2024
Abstract
SARS-CoV-2 virus variants of concern (VOCs) have rapidly changed their transmissibility and pathogenicity primarily through mutations in the structural proteins. Herein, we present molecular details with dynamics of the ferritin nanocages stitched with synthetic chimeras displaying the Spike receptor binding domains (RBDs). Our
[...] Read more.
SARS-CoV-2 virus variants of concern (VOCs) have rapidly changed their transmissibility and pathogenicity primarily through mutations in the structural proteins. Herein, we present molecular details with dynamics of the ferritin nanocages stitched with synthetic chimeras displaying the Spike receptor binding domains (RBDs). Our findings demonstrated the potential usage of ferritin-based vaccines that may effectively inhibit viral entry by blocking the Spike–ACE2 network and may induce cross-protective antibody responses. Taking the nanocage constructs into consideration, we evaluated the effects of variants on the docked interface of the SARS-CoV-2 Spike RBD with the ACE2 (angiotensin-converting enzyme 2) host cell receptor and neutralizing antibodies (Abs). Investigating the VOCs revealed that most of the mutations reported a possibly reduced structural stability within the Spike RBD domain. Point mutations have moderate or no effect for VVH-72, CR3022, and S309 Abs when bound with the Spike RBD, whereas a significant effect was observed for B38, CB6, and m396 over the surface of the H-ferritin nanocage. In addition to providing useful therapeutic approaches against COVID-19 (coronavirus disease 2019), these structural details can also be used to fight future coronavirus outbreaks.
Full article
(This article belongs to the Special Issue Influence of Natural and/or Vaccine Immunity on the Dynamics of SARS-CoV-2)
►▼
Show Figures
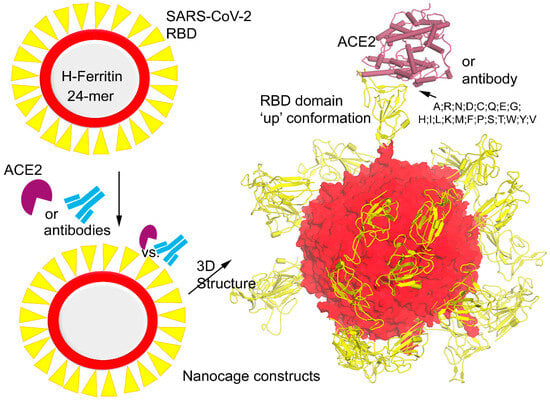
Figure 1
Open AccessReview
Vaccine Hesitancy among Immigrants: A Narrative Review of Challenges, Opportunities, and Lessons Learned
by
Jason Wong, Crystal Lao, Giancarlo Dino, Roujina Donyaei, Rachel Lui and Jennie Huynh
Vaccines 2024, 12(5), 445; https://doi.org/10.3390/vaccines12050445 - 23 Apr 2024
Abstract
(1) Background: Vaccination reluctance is a major worldwide public health concern as it poses threats of disease outbreaks and strains on healthcare systems. While some studies have examined vaccine uptake within specific countries, few provide an overview of the barriers and trends among
[...] Read more.
(1) Background: Vaccination reluctance is a major worldwide public health concern as it poses threats of disease outbreaks and strains on healthcare systems. While some studies have examined vaccine uptake within specific countries, few provide an overview of the barriers and trends among migrant groups. To fill this knowledge gap, this narrative review analyzes immunization patterns and vaccine hesitancy among immigrant populations. (2) Methods: Four researchers independently evaluated the quality and bias risk of the 18 identified articles using validated critical appraisal tools. (3) Results: Most studies focused on vaccine hesitancy among migrants in the United States and Canada, with a higher COVID-19 vaccine reluctance than native-born residents. Contributing factors to this hesitancy include demographics, cultural views, obstacles to healthcare access, financial hardship, and distrust in health policies. Additionally, immigrants in North America and Europe face unfair vaccine challenges due to misinformation, safety concerns, personal perspectives, language barriers, immigration status, and restricted healthcare access. (4) Conclusions: Tailored vaccine education programs and outreach campaigns sensitive to immigrants’ diversity should be developed to address this issue. It is also important to investigate community-specific obstacles and assess the long-term sustainability of current efforts to promote vaccination among marginalized migrant groups. Further research into global immunization disparities among immigrant populations is crucial.
Full article
(This article belongs to the Special Issue Vaccine Hesitancy and Acceptance, Trends and Future Prospects for Public Health)
►▼
Show Figures
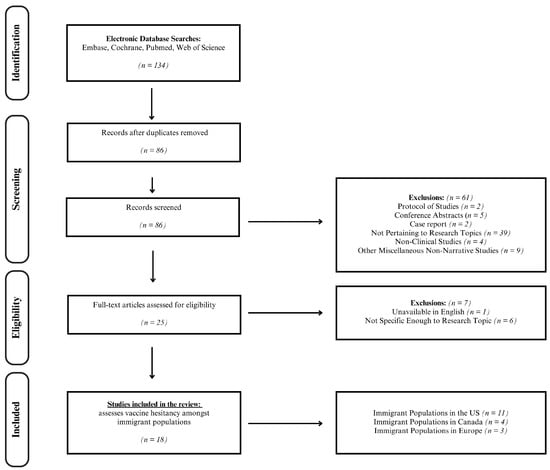
Figure 1
Open AccessArticle
COVID-19 Vaccine Hesitancy and Associated Oral Cholera Vaccine Hesitancy in a Cholera-Endemic Country: A Community-Based Cross-Sectional Study in the Democratic Republic of Congo
by
Arsene Daniel Nyalundja, Patrick Musole Bugeme, Alain Balola Ntaboba, Victoire Urbain Hatu’m, Guillaume Shamamba Ashuza, Jacques Lukenze Tamuzi, Duduzile Ndwandwe, Chinwe Iwu-Jaja, Charles Shey Wiysonge and Patrick D. M. C. Katoto
Vaccines 2024, 12(4), 444; https://doi.org/10.3390/vaccines12040444 - 22 Apr 2024
Abstract
COVID-19 vaccine hesitancy and its enablers shape community uptake of non-covid vaccines such as the oral cholera vaccine (OCV) in the post-COVID-19 era. This study assessed the impact of COVID-19 vaccine hesitancy and its drivers on OCV hesitancy in a cholera-endemic region of
[...] Read more.
COVID-19 vaccine hesitancy and its enablers shape community uptake of non-covid vaccines such as the oral cholera vaccine (OCV) in the post-COVID-19 era. This study assessed the impact of COVID-19 vaccine hesitancy and its drivers on OCV hesitancy in a cholera-endemic region of the Democratic Republic of Congo. We conducted a community-based survey in Bukavu. The survey included demographics, intention to take OCV and COVID-19 vaccines, reasons for COVID-19 hesitancy, and thoughts and feelings about COVID-19 vaccines. Poisson regression analyses were performed. Of the 1708 respondents, 84.66% and 77.57% were hesitant to OCV alone and to both OCV and COVID-19, respectively. Hesitancy to COVID-19 vaccines rose OCV hesitancy by 12% (crude prevalence ratio, [cPR] = 1.12, 95%CI [1.03–1.21]). Independent predictors of OCV hesitancy were living in a semi-urban area (adjusted prevalence ratio [aPR] = 1.10, 95%CI [1.03–1.12]), religious refusal of vaccines (aPR = 1.06, 95%CI [1.02–1.12]), concerns about vaccine safety (aPR = 1.05, 95%CI [1.01–1.11]) and adverse effects (aPR = 1.06, 95%CI [1.01–1.12]), as well as poor vaccine literacy (aPR = 1.07, 95%CI [1.01–1.14]). Interestingly, the belief in COVID-19 vaccine effectiveness reduced OCV hesitancy by 24% (aPR = 0.76, 95%CI [0.62–0.93]). COVID-19 vaccine hesitancy and its drivers exhibited a significant domino effect on OCV uptake. Addressing vaccine hesitancy through community-based health literacy and trust-building interventions would likely improve the introduction of novel non-COVID-19 vaccines in the post-COVID-19 era.
Full article
(This article belongs to the Special Issue 50 Years of Immunization—Steps Forward)
Open AccessArticle
Characterization of the Protective Cellular Immune Response in Pigs Immunized Intradermally with the Live Attenuated African Swine Fever Virus (ASFV) Lv17/WB/Rie1
by
Miriam Pedrera, Alejandro Soler, Alicia Simón, Nadia Casado, Covadonga Pérez, María A. García-Casado, Paloma Fernández-Pacheco, Pedro J. Sánchez-Cordón, Marisa Arias and Carmina Gallardo
Vaccines 2024, 12(4), 443; https://doi.org/10.3390/vaccines12040443 - 20 Apr 2024
Abstract
Candidate vaccines against African swine fever virus (ASFV) based on naturally attenuated or genetically modified viruses have the potential to generate protective immune responses, although there is no consensus on what defines a protective immune response against ASFV. Studies, especially in sensitive host
[...] Read more.
Candidate vaccines against African swine fever virus (ASFV) based on naturally attenuated or genetically modified viruses have the potential to generate protective immune responses, although there is no consensus on what defines a protective immune response against ASFV. Studies, especially in sensitive host species and focused on unravelling protective mechanisms, will contribute to the development of safer and more effective vaccines. The present study provides a detailed analysis of phenotypic and functional data on cellular responses induced by intradermal immunization and subsequent boosting of domestic pigs with the naturally attenuated field strain Lv17/WB/Rie1, as well as the mechanisms underlying protection against intramuscular challenge with the virulent genotype II Armenia/07 strain. The transient increase in IL-8 and IL-10 in serum observed after immunization might be correlated with survival. Protection was also associated with a robust ASFV-specific polyfunctional memory T-cell response, where CD4CD8 and CD8 T cells were identified as the main cellular sources of virus-specific IFNγ and TNFα. In parallel with the cytokine response, these T-cell subsets also showed specific cytotoxic activity as evidenced by the increased expression of the CD107a degranulation marker. Along with virus-specific multifunctional CD4CD8 and CD8 T-cell responses, the increased levels of antigen experienced in cytotoxic CD4 T cells observed after the challenge in immunized pigs might also contribute to controlling virulent infection by killing mechanisms targeting infected antigen-presenting cells. Future studies should elucidate whether the memory T-cell responses evidenced in the present study persist and provide long-term protection against further ASFV infections.
Full article
(This article belongs to the Special Issue Diagnosis and Control of African Swine Fever Virus (ASFV) Infection)
►▼
Show Figures
Figure 1
Open AccessArticle
Anti-Herpes Zoster Vaccination of Fragile Patients in Hospital Setting: A Nudge Intervention in Italy
by
Francesco De Caro, Francesca Malatesta, Nadia Pecoraro, Mario Capunzo, Luna Carpinelli, Simona Caruccio, Giuseppina Cersosimo, Maria Costantino, Claudio Giordano, Walter Longanella, Vincenzo Patella, Arcangelo Saggese Tozzi, Giulia Savarese, Pio Sinopoli, Emilia Anna Vozzella and Giuseppina Moccia
Vaccines 2024, 12(4), 442; https://doi.org/10.3390/vaccines12040442 - 19 Apr 2024
Abstract
Background: A nudge intervention against Herpes Zoster, created and implemented in Italy, is presented in order to administer the Shingrix vaccine on a sample of frail patients, as required by the National Prevention Plan. Individual and contextual factors associated with vaccine adherence were
[...] Read more.
Background: A nudge intervention against Herpes Zoster, created and implemented in Italy, is presented in order to administer the Shingrix vaccine on a sample of frail patients, as required by the National Prevention Plan. Individual and contextual factors associated with vaccine adherence were investigated. Method: 300 frail adult subjects underwent a full vaccine cycle with recombinant-Shingrix vaccine (RZV vaccine). Hospital Presidia of the Salerno University Hospital Authority, a Hospital Presidium of the Salerno Local Health Authority, and the Public Health Laboratory of the University of Salerno (Campania) participated in the intervention. An ad hoc questionnaire was administered with the following scales: EQ-5D, PSS-10, MSPSS, and representations of HZ and its consequences. Results: Some variables, such as peer support, doctor–patient relationship, level of education, and perception of health, are important in vaccine adherence and information processing. The following factors emerged from the factor analysis: Trust in collective knowledge and collective responsibility (F1); beliefs about virus risk and vaccine function (F2); information about virus and symptomatology (F3); and vaccine distrust (F4). Factor 4 correlates negatively with social support indices (R = −0.363; p < 0.001). There is a significant relationship between factor 3 and satisfaction with national information campaigns (F = 3.376; gdl = 5; p-value = 0.006). Conclusions: Future vaccination campaigns should be built with the aim of personalizing information and developing contextualized strategies, starting from understanding the stakeholders involved, cultural contexts, and organizational settings.
Full article
(This article belongs to the Special Issue Vaccination Uptake and Public Health)
Open AccessArticle
The Papain-like Protease Domain of Severe Acute Respiratory Syndrome Coronavirus 2 Conjugated with Human Beta-Defensin 2 and Co1 Induces Mucosal and Systemic Immune Responses against the Virus
by
Byeol-Hee Cho, Ju Kim and Yong-Suk Jang
Vaccines 2024, 12(4), 441; https://doi.org/10.3390/vaccines12040441 - 19 Apr 2024
Abstract
Most of the licensed vaccines against SARS-CoV-2 target spike proteins to induce viral neutralizing antibodies. However, currently prevalent SARS-CoV-2 variants contain many mutations, especially in their spike proteins. The development of vaccine antigens with conserved sequences that cross-react with variants of SARS-CoV-2 is
[...] Read more.
Most of the licensed vaccines against SARS-CoV-2 target spike proteins to induce viral neutralizing antibodies. However, currently prevalent SARS-CoV-2 variants contain many mutations, especially in their spike proteins. The development of vaccine antigens with conserved sequences that cross-react with variants of SARS-CoV-2 is needed to effectively defend against SARS-CoV-2 infection. Given that viral infection is initiated in the respiratory mucosa, strengthening the mucosal immune response would provide effective protection. We constructed a mucosal vaccine antigen using the papain-like protease (PLpro) domain of non-structural protein 3 of SARS-CoV-2. To potentiate the mucosal immune response, PLpro was combined with human beta-defensin 2, an antimicrobial peptide with mucosal immune adjuvant activity, and Co1, an M-cell-targeting ligand. Intranasal administration of the recombinant PLpro antigen conjugate into C57BL/6 and hACE2 knock-in (KI) mice induced antigen-specific T-cell and antibody responses with complement-dependent cytotoxic activity. Viral challenge experiments using the Wuhan and Delta strains of SARS-CoV-2 provided further evidence that immunized hACE2 KI mice were protected against viral challenge infections. Our study shows that PLpro is a useful candidate vaccine antigen against SARS-CoV-2 infection and that the inclusion of human beta-defensin 2 and Co1 in the recombinant construct may enhance the efficacy of the vaccine.
Full article
(This article belongs to the Section Vaccine Adjuvants)
►▼
Show Figures
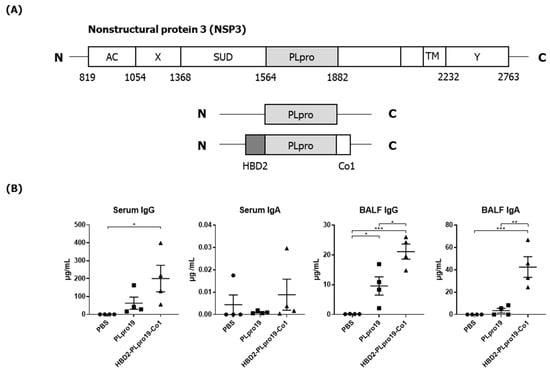
Figure 1
Open AccessArticle
Courage in Decision Making: A Mixed-Methods Study of COVID-19 Vaccine Uptake in Women of Reproductive Age in the U.K.
by
Laura A. Magee, Julia R. Brown, Vicky Bowyer, Gillian Horgan, Harriet Boulding, Asma Khalil, Nathan J. Cheetham, Nicholas R. Harvey, COVID Symptom Study Biobank Consortium, RESILIENT Study Group, Hiten D. Mistry, Carole Sudre, Sergio A. Silverio, Peter von Dadelszen and Emma L. Duncan
Vaccines 2024, 12(4), 440; https://doi.org/10.3390/vaccines12040440 - 18 Apr 2024
Abstract
COVID-19 vaccination rates are lower in women of reproductive age (WRA), including pregnant/postpartum women, despite their poorer COVID-19-related outcomes. We evaluated the vaccination experiences of 3568 U.K. WRA, including 1983 women (55.6%) experiencing a pandemic pregnancy, recruited through the ZOE COVID Symptom Study
[...] Read more.
COVID-19 vaccination rates are lower in women of reproductive age (WRA), including pregnant/postpartum women, despite their poorer COVID-19-related outcomes. We evaluated the vaccination experiences of 3568 U.K. WRA, including 1983 women (55.6%) experiencing a pandemic pregnancy, recruited through the ZOE COVID Symptom Study app. Two staggered online questionnaires (Oct–Dec 2021: 3453 responders; Aug–Sept 2022: 2129 responders) assessed reproductive status, COVID-19 status, vaccination, and attitudes for/against vaccination. Descriptive analyses included vaccination type(s), timing relative to age-based eligibility and reproductive status, vaccination delay (first vaccination >28 days from eligibility), and rationale, with content analysis of free-text comments. Most responders (3392/3453, 98.2%) were vaccinated by Dec 2021, motivated by altruism, vaccination supportiveness in general, low risk, and COVID-19 concerns. Few declined vaccination (by Sept/2022: 20/2129, 1.0%), citing risks (pregnancy-specific and longer-term), pre-existing immunity, and personal/philosophical reasons. Few women delayed vaccination, although pregnant/postpartum women (vs. other WRA) received vaccination later (median 3 vs. 0 days after eligibility, p < 0.0001). Despite high uptake, concerns included adverse effects, misinformation (including from healthcare providers), ever-changing government advice, and complex decision making. In summary, most women in this large WRA cohort were promptly vaccinated, including pregnant/post-partum women. Altruism and community benefit superseded personal benefit as reasons for vaccination. Nevertheless, responders experienced angst and received vaccine-related misinformation and discouragement. These findings should inform vaccination strategies in WRA.
Full article
(This article belongs to the Special Issue COVID-19 Vaccination with Public Health Perspective: Prevention and Control Strategies)
Open AccessReview
Hepatitis B Vaccine: Four Decades on
by
Maria Mironova and Marc G. Ghany
Vaccines 2024, 12(4), 439; https://doi.org/10.3390/vaccines12040439 - 18 Apr 2024
Abstract
Hepatitis B virus is a substantial contributor to cirrhosis and hepatocellular carcinoma (HCC) globally. Vaccination is the most effective method for prevention of hepatitis B and its associated morbidity and mortality, and the only method to prevent infection with hepatitis D virus. The
[...] Read more.
Hepatitis B virus is a substantial contributor to cirrhosis and hepatocellular carcinoma (HCC) globally. Vaccination is the most effective method for prevention of hepatitis B and its associated morbidity and mortality, and the only method to prevent infection with hepatitis D virus. The hepatitis B vaccine has been used worldwide for more than four decades; it is available in a single- or triple-antigen form and in combination with vaccines against other infections. Introduction of the vaccine and administration at birth led to sustained decline in mother-to-child transmission, chronic hepatitis B, and HCC, however, global birth dose coverage remains suboptimal. In this review we will discuss different hepatitis B vaccine formulations and schedules, vaccination guidelines, durability of the response, and vaccine escape mutants, as well as the clinical and economic benefits of vaccination.
Full article
(This article belongs to the Special Issue Efficacy, Safety, and Immunogenicity of Hepatitis B Vaccines)
►▼
Show Figures
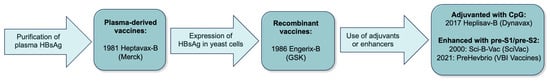
Figure 1
Open AccessArticle
Using a Dynamic Model to Estimate the Cost-Effectiveness of HPV Vaccination in Iran
by
Arnold Hagens, Albertus Constantijn Sloof and Roksana Janghorban
Vaccines 2024, 12(4), 438; https://doi.org/10.3390/vaccines12040438 - 18 Apr 2024
Abstract
This study aimed to determine the cost-effectiveness of vaccination against HPV. An age–sex structured dynamic disease transmission model was created to estimate the spread of HPV and the HPV-related incidence of cervical cancer (CC) in Iran. Sixteen age groups of men and women
[...] Read more.
This study aimed to determine the cost-effectiveness of vaccination against HPV. An age–sex structured dynamic disease transmission model was created to estimate the spread of HPV and the HPV-related incidence of cervical cancer (CC) in Iran. Sixteen age groups of men and women were incorporated to reflect the differences in sexual preferences, vaccination uptake, and disease-related outcomes. Three scenarios were evaluated by using an Incremental Cost-Effectiveness Ratio (ICER) with gained quality-adjusted life years (QALYs). ICER values below one gross domestic product (GDP) per capita are evaluated as highly cost-effective. Vaccination reduces the number of infections and CC-related mortality. Over time, the vaccinated group ages and older age groups experience protection. An initial investment is required and savings in treatment spending reduce the impact over time. Vaccinating girls only was found to be cost-effective, with an ICER close to once the GDP per capita. Vaccinating both sexes was shown to be less cost-effective compared to girls only, and vaccinating boys only was not found to be cost-effective, with an ICER between once and three times, and greater than three times the GDP per capita, respectively. The estimates are conservative since societal cost-saving and the impact of other HPV-related illnesses were not considered and would likely reduce the ICERs.
Full article
(This article belongs to the Special Issue HPV in the Young Population: New Perspectives in Clinical Practice from Vaccination to Screening)
►▼
Show Figures
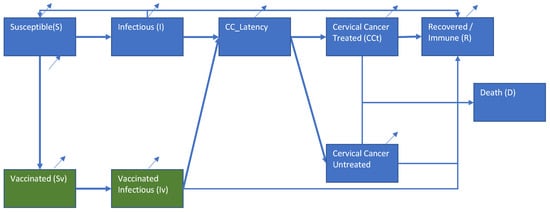
Figure 1
Open AccessArticle
Analysis of Protection and Immune Response against Teladorsagia circumcincta in Goats Immunised with Thiol-Binding Proteins from Adult Worms
by
Leire Ortega, Jessica Quesada, Antonio Ruiz, Magnolia María Conde-Felipe, Otilia Ferrer, María del Carmen Muñoz, José Adrián Molina, Francisco Rodríguez and José Manuel Molina
Vaccines 2024, 12(4), 437; https://doi.org/10.3390/vaccines12040437 - 18 Apr 2024
Abstract
In view of the increasing occurrence of anthelmintic-resistant strains of gastrointestinal nematodes in ruminants, various alternative control strategies have been investigated, such as those based on the induction of protective immune responses by immunisation with parasite antigens. In this study, the protective activity
[...] Read more.
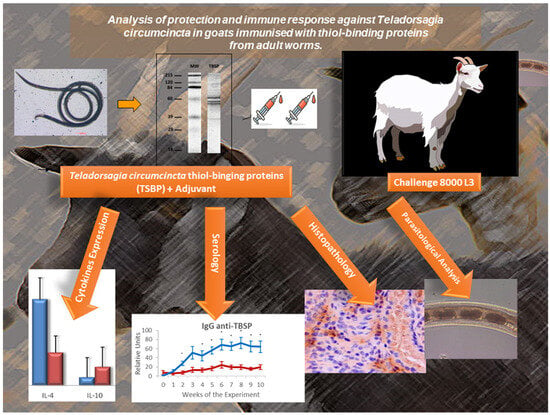
In view of the increasing occurrence of anthelmintic-resistant strains of gastrointestinal nematodes in ruminants, various alternative control strategies have been investigated, such as those based on the induction of protective immune responses by immunisation with parasite antigens. In this study, the protective activity of somatic antigens from adult worms of Teladorsagia circumcincta purified by affinity chromatography on thiol-sepharose was analysed in goats. After challenge, the enriched products induced a slight reduction in the cumulative faecal egg counts (21%) and in the number of worms (23.3%), with a greater effect on female worms, which also showed a reduction in parameters related to their fertility. These parasitological findings were associated with a Th2 immune response, with a prominent local humoral response and an eosinophilic infiltrate in the gastric mucosa (negatively associated with the fertility of female worms and the number of worms, respectively), as well as an infiltration of MCHII+, CD4+, IgG+ and IgA+ cells. However, several analyses showed an increase in CD8+ cells in the mucosa, as well as IL-2 expression in the gastric lymph nodes, which may have been associated with inhibition of protective responses or with the development of mixed Th1/Th2 responses, a finding that should be analysed in future studies.
Full article
(This article belongs to the Special Issue Parasitic Infections: Therapy for Host Immunity and Vaccination)
►▼
Show Figures
Graphical abstract

Journal Menu
► ▼ Journal Menu-
- Vaccines Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Diseases, IJMS, Microbiology Research, Pathogens, Vaccines
Advances in Human Pathogen Control—a 21st Century Challenge 2.0
Topic Editors: Jorge H. Leitão, Nitin Amdare, Joana R FelicianoDeadline: 30 June 2024
Topic in
Cells, Diseases, Healthcare, IJMS, Vaccines
Inflammation: The Cause of all Diseases 2.0
Topic Editors: Vasso Apostolopoulos, Jack Feehan, Vivek P. ChavdaDeadline: 31 July 2024
Topic in
Biomedicines, JCM, Pathogens, Vaccines, Viruses
Discovery and Development of Monkeypox Disease Treatments
Topic Editors: Mohd Imran, Ali A. RabaanDeadline: 31 August 2024
Topic in
Brain Sciences, Clinics and Practice, COVID, Life, Vaccines, Viruses
Multifaceted Efforts from Basic Research to Clinical Practice in Controlling COVID-19 Disease
Topic Editors: Yih-Horng Shiao, Rashi OjhaDeadline: 30 September 2024

Conferences
Special Issues
Special Issue in
Vaccines
Public Psychobehavioral Responses towards Vaccination
Guest Editor: Li Ping WongDeadline: 30 April 2024
Special Issue in
Vaccines
Novel Vaccines for Infectious Pathogens
Guest Editors: Veerupaxagouda Patil, Dhruv DesaiDeadline: 20 May 2024
Special Issue in
Vaccines
COVID-19 Vaccine Acceptance and Uptake: Insights from Behavioural and Social Sciences
Guest Editor: Christian NapoliDeadline: 31 May 2024
Special Issue in
Vaccines
SARS-CoV-2 Variants, Vaccines, and Immune Responses
Guest Editor: Gianni Gori SavelliniDeadline: 15 June 2024
Topical Collections
Topical Collection in
Vaccines
COVID-19 Vaccine Hesitancy: Correlates and Interventions
Collection Editors: Manoj Sharma, Kavita Batra
Topical Collection in
Vaccines
Topic Advisory Panel Members’ Collection Series: Immunization and Vaccines for Infectious Diseases
Collection Editors: Shumaila Hanif, Ravinder Kumar
Topical Collection in
Vaccines
Research on Monoclonal Antibodies and Antibody Engineering
Collection Editor: Tatsuya Yamazaki






